Based on 268+ reviews

150,000+
USERS
116
COUNTRIES
7,400+
MAINTENANCE TEAMS HELPED
3.4 Million
MACHINES FIXED
eMaint CMMS goes beyond traditional maintenance software by also serving as a maintenance & reliability compliance hub.
Production shuts down when audits go wrong — and for some businesses, that can mean a loss of as much as $100,000 per hour of downtime. A CMMS designed with compliance in mind has built-in functionalities and guardrails to optimize your compliance.
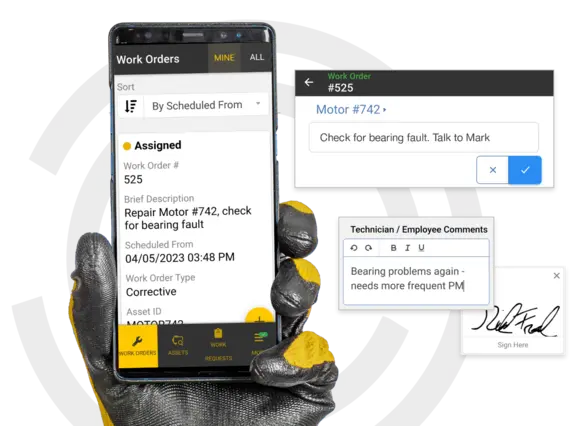
Standardize a Winning Maintenance Strategy for Compliance
Automate compliant processes with workflows, store key procedures and documents, and move work orders through levels of approval.
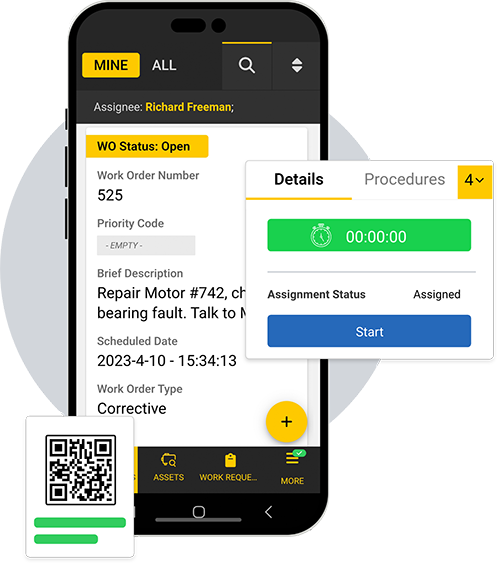
You don’t have time to supervise every work order — let eMaint guide your team through compliant processes
Automate best practices across sites
eMaint workflows give you the power to guide work orders through your standardized processes. eMaint is a multi-site CMMS, so you can scale your strategy across multiple teams and languages.
Your global compliance hub
House and reference regulatory documents and standards. Build your procedures into work order templates. Indicate the compliance history of key assets and specific instructions in asset notes.
Route work orders through safety, quality, sanitation
Establish controls over maintenance work with workflows that automatically route work orders through supervisors. Route work orders to key safety, quality, or sanitation personnel to gain oversight and approval.
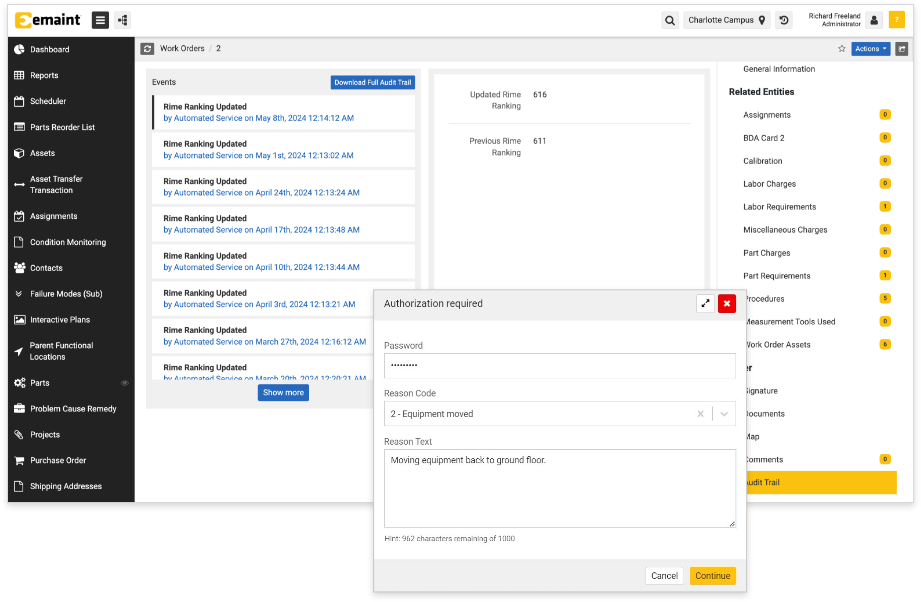
Everything is Tracked with eMaint’s Audit Trail & E-Signatures
eMaint records user actions in an easily-accessible audit trail and offers digital signatures to safeguard key actions.
Your team’s actions across work orders, parts, assets, and beyond — always just a few clicks away
Quickly demonstrate compliance
The eMaint audit trail is easily accessible in records like work orders or assets. Click in to show asset maintenance history and proof of work order completion.
Comprehensive audit tracking
You can find detailed, timestamped records of user actions and changes by clicking into the audit trail for a record. The audit trail is secure and protected from user edits. Trust in a system designed with FDA 21 CFR Part 11 and EudraLex Vol. 4 in mind.
Control key actions with e-signatures
Require digital signatures for important actions or field edits. E-signatures are password protected to ensure validity. You can also capture a reason code and/or description for each change during the e-signature process.
Simple, Flexible Audit Compliance Reporting
Build reports and dashboards that streamline audit compliance.
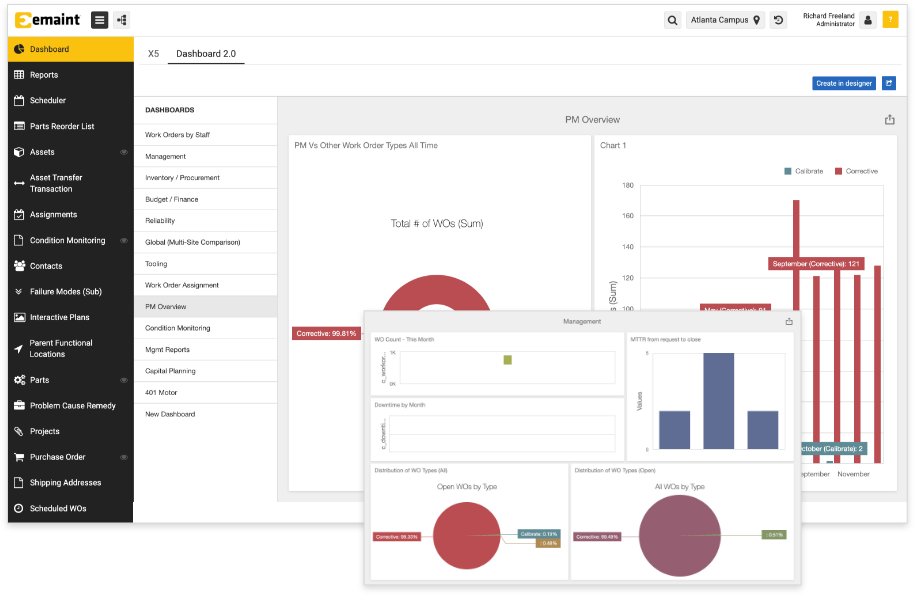
Get auditors in and out with powerful reports and dashboards
We’re here to help
Our team will design reports and dashboards for you that give instant visibility into your compliant practices. Work with our CMMS experts to determine what reporting, for both internal and external audiences, you need to simplify your workday.
Make audits painless
Design dashboards in a few clicks that demonstrate PM compliance and on-track PM completion rates. Automate reports to generate with key data for regulatory audits, from OSHA to SQF and IATF.
Gain powerful, real-time visibility
How many open work orders does your team have? What’s the asset health status of your critical equipment? Are your running low on key spare parts? eMaint gives you real-time visibility for managing and monitoring your team, assets, parts, and more.

