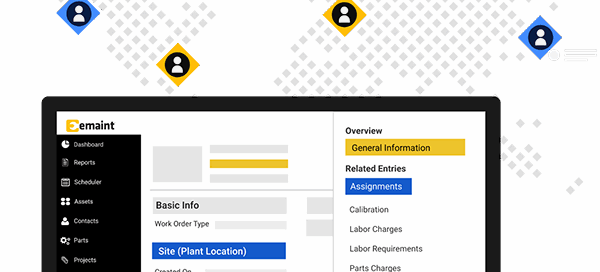
Why cold chain reliability starts with maintenance: What the industry’s rapid growth means for compliance managers
UNICEF is expected to deliver approximately three billion vaccine doses to children around the world this year, each one dependent on a carefully managed cold [...]