Software de Manutenção das Ciências da Vida para Conformidade Regulamentar
Simplificar a conformidade e calibração com eMaint CMMS para as ciências da vida
Os governos regulamentam rigorosamente as empresas das ciências da vida. Os profissionais de manutenção e operações devem cumprir as leis locais, estaduais, federais, e internacionais. Com tantos regulamentos, não é de admirar que muitas organizações achem as normas de informação e auditoria confusas e incómodas. É aí que entra o software de sistemas de gestão de manutenção computorizados (CMMS ).
Como uma das melhores soluções disponíveis em ciências da vida, o eMaint CMMS simplifica
a conformidade, criando um rasto digital em papel para os inspectores
a conformidade, criando um rasto digital em papel para os inspectores
Com documentação digital, pode facilmente partilhar provas de conformidade com auditores. Não só as organizações de ciências da vida
provarão mais facilmente a conformidade com registos e assinaturas digitais, como também experimentarão uma maior fiabilidade operacional.
Os 5 principais benefícios do CMMS para as Ciências da Vida
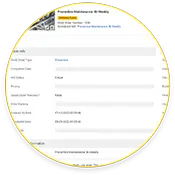
Criação de pista de auditoria
As equipas de manutenção que utilizam eMaint CMMS criam pistas de conformidade de ponta a ponta para 21 auditorias CFR Part 11, incluindo históricos de bens, conclusão de ordens de trabalho, e assinaturas electrónicas.
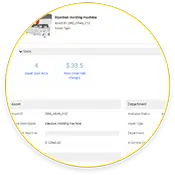
Racionalização da Saúde e História dos Bens
Não só o eMaint CMMS armazena históricos de encomendas de bens, como também inclui em tempo real e histórico monitoramento de condição dados.
Calibração Automática e PMs
As acções de manutenção preventiva de conformidade e calibração podem ser programadas ou automatizadas utilizando o eMaint CMMS.
Seguimento de Trabalho e Inventário
À medida que as ordens de trabalho são completadas, o acompanhamento automático dos níveis de trabalho e de inventário ajuda os gestores a planear e programar as futuras PMs e as ordens de reabastecimento.
Tomar decisões financeiras mais inteligentes
Com registos adequados, a liderança pode tomar decisões mais informadas sobre reparação/substituição de bens, implementações IIoT, e manutenção centrada na fiabilidade.

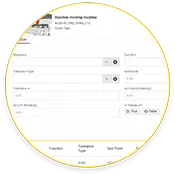
Calibração
Programar ordens de trabalho de calibração para verificar a precisão das medições monitoramento de condição .
Simplificar as auditorias de conformidade, automatizando as ordens de trabalho de calibração e rastreando os horários das ordens de trabalho de manutenção preventiva (PM).
Aumentar a precisão através da simplificação da manutenção de registos com actividades digitalizadas, incluindo calibração, testes, e medições.
Reduzir as dores de cabeça de auditoria de conformidade, eliminando os aglutinantes volumosos e reduzindo os erros de introdução de dados nos registos.
Bens que precisam de calibração
Centrífugas
Elementos de Pressão
Indicadores de pressão
Balança
Temperatura
Pressão
Métricas de fluxo
Pipetas
Incubadoras
Verticais das Ciências da Vida que beneficiam de um CMMS
Agricultura, Pescas, & Alimentação
Anatomia & Morfologia
Ciências Comportamentais
Biologia
Bioquímica
Biotecnologia
Biofísica
Botânica
Citologia
Ecologia e Ambiente
Entomologia
Silvicultura
Genética e Hereditariedade
Imunologia
Microbiologia
Micologia
Parasitologia
Farmacêuticos
Farmacologia
Fisiologia
Ciências vegetais
Toxicologia
Ciências Veterinárias
Virologia
Zoologia
E mais